What is chronic kidney disease (CKD)?
Chronic kidney disease (CKD) is a progressive condition where the kidneys gradually lose function [1]. It is characterized by a high morbidity and mortality rate, and it is predicted to be the 5th leading cause of death globally by 2040 [2]. It commonly affects the general adult population, but people with hypertension or diabetes are increasingly at risk.
The damage caused by CKD is usually irreversible. As there is no cure, treatment aims to mitigate the progression of the disease through pharmacological (CKD-targeted drugs and kidney-specific pharmacological interventions) and non-pharmacological (dietary and lifestyle changes) means. However, monitoring CKD presents many challenges, which will be discussed below.
Detecting CKD in its early stages
Detection of CKD in its early stages is challenging and uncommon [3]. Early diagnosis is critical for preventing the progression to end-stage renal disease, but the initial phases of the disease are usually asymptomatic, leading to lower diagnosis rates [3,4]. Furthermore, the human eye’s limitations during manual assessments can lead to a semi-crude identification of abnormal changes in kidney structures. This often results in early-stage abnormalities being overlooked, even if patients provide the samples.
AI holds the potential to improve early detection through advanced analysis of digital pathology slides with high precision. This technology can identify subtle changes and patterns that might be missed by human eyes, ensuring a more accurate and timely diagnosis [2,5]. Additionally, AI-powered analysis can support biomarker research and discovery, where machine learning algorithms analyze vast datasets to pinpoint early indicators of CKD, contributing to higher detection rates and improved patient outcomes [2]. Utilizing AI kidney disease models for biopsy analysis can ensure that even slight deviations from normal kidney function are flagged and addressed promptly, potentially preventing disease progression.
Monitoring CKD progression with AI assistance
Due to the incurable nature of CKD, closely monitoring its progression is essential to prevent further complications and delay the most debilitating symptoms [6]. Comparative analysis between historical and current slides is crucial for assessing treatment effectiveness and prognosis. Making an accurate comparison is difficult without standardized procedures for assessing the progression of, for example, fibrotic structures.
Deep-learning technology can facilitate these comparisons by providing laboratories with standardized criteria for slide assessments. AI kidney disease solutions offer the possibility of evaluating the fibrotic area with accurate percentages, unlike the broader Banff ranges used in manual assessments, which are prone to high inter-observer variability [7,12,13]. This precision may be vital in assessing the effectiveness of drugs and the progression of the disease. Additionally, AI-powered digital pathology can incorporate personalized quantitative imaging biomarkers, which provide objective, measurable data points regarding the extent of kidney damage [2]. These biomarkers can be tracked over time, offering a detailed and precise picture of disease progression that informs better clinical decisions.
Machine learning algorithms can quickly and consistently analyze large amounts of information, establishing laboratory procedures to provide reproducible results without significant human resources. By reducing the workload for pathologists, AI enables the processing of more samples and allows pathologists to focus on higher-level diagnostics [7].
End-stage renal disease (ESRD): AI assistance in transplantation biopsies
With the progression of chronic kidney disease, renal functions keep declining. The dangerous accumulation of fluids, electrolytes, and waste in the kidneys builds up in the organism, leading to a life-threatening condition. During those late stages, there is a need for more extreme treatment methods that can include transplantation or dialysis. It is crucial to monitor the effectiveness of these treatments to ensure patient safety and health. After transplantation, it is especially important to carefully monitor the post-transplantation kidney function to ensure the success of the treatment [9]. In cases of declining functionality, biopsies are taken to assess the state of the kidney.
Advanced machine-learning techniques can provide objective and reproducible quantifications of kidney tissue features that can improve the diagnostics of kidney transplants [10,11]. Typically, transplantation assessments involve evaluating outcomes like kidney functions over the years and patient survival. Additionally, it can be evaluated through semi-crude quantification of the kidney damage using Banff scores [12]. However, with the assistance of AI kidney disease algorithms, researchers might use surrogate end-points that can be measured quicker, such as the detection of tissue scarring or fibrosis, enabling better treatment plans and a more accurate prognosis [11].
The future of CKD management: AI’s role in precision medicine
AI has transformative potential in managing CKD from diagnosis through monitoring and treatment management. Computational pathology can provide rapid, standardized, and accurate assessments of disease progression and improve treatment effectiveness evaluations. This comprehensive approach enables consistent, efficient, and timely CKD management, fundamentally transforming traditional practices.
Ultimately, integrating AI-powered precision pathology solutions into CKD management workflows not only enhances diagnostic and monitoring accuracy but also paves the way for personalized medicine [2]. By tailoring treatment plans based on precise, data-driven insights, healthcare providers can improve patient outcomes and quality of life.
The future of AI in digital pathology promises further advancements. With ongoing research and development, AI kidney disease models will likely incorporate even more sophisticated capabilities, which could revolutionize how CKD is managed and treated [2].
Aiosyn’s AI kidney disease solutions
At Aiosyn, we recognize the importance of developing cutting-edge solutions to improve kidney pathology assessments. Our NephroPath platform provides detailed evaluations of histological biomarkers in pre-clinical and clinical kidney samples*. By leveraging advanced AI algorithms, we offer both standard tissue quantification and custom analyses tailored to your needs.
Contact us to learn more about how our AI-based kidney image analysis services can support your lab.
* The NephroPath platform is currently for Research Use Only (RUO) and should not be used for diagnostic procedures.
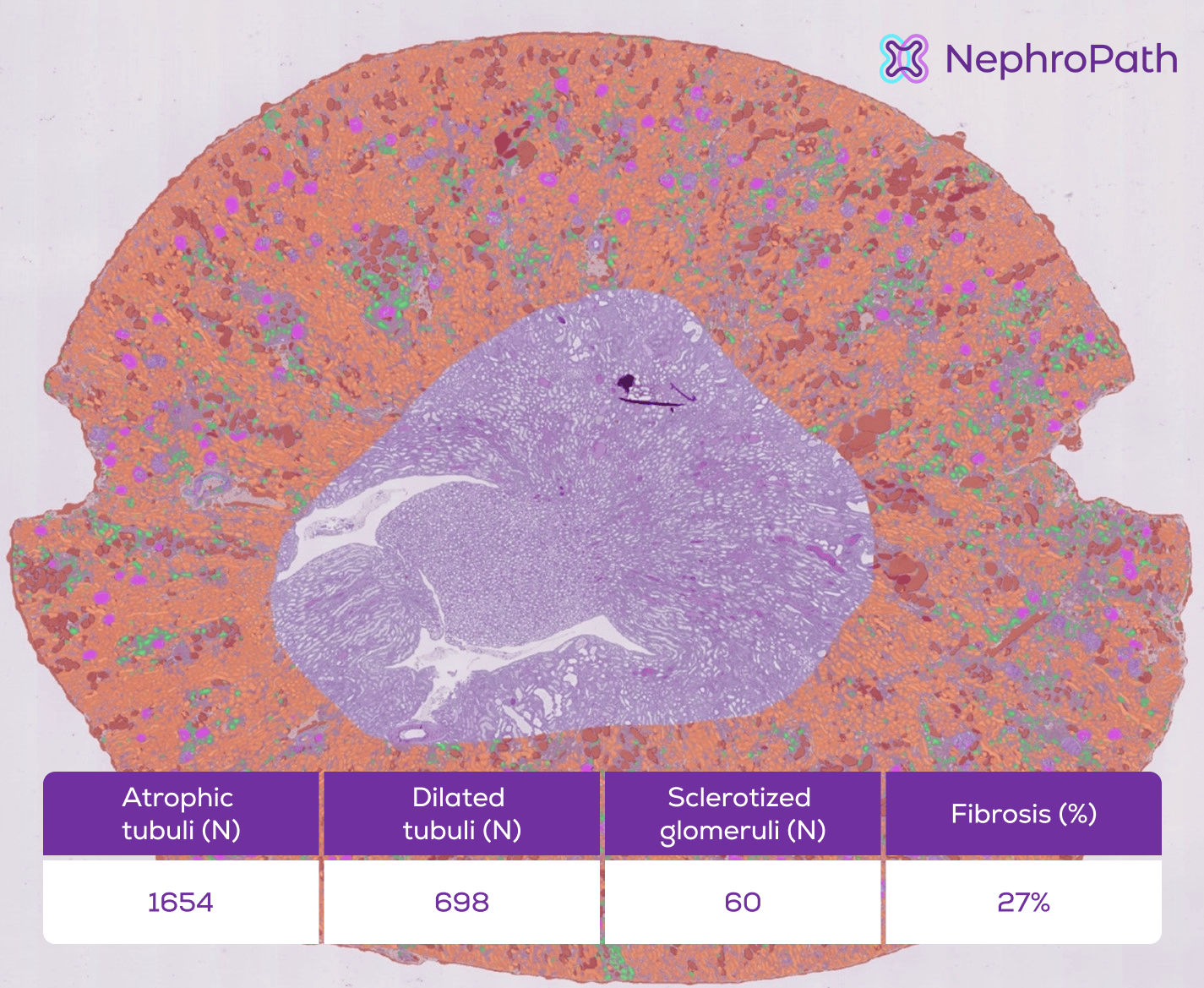
Figure 1. Illustration of the NephroPath platform’s capabilities in performing automated quantification and multi-class prediction on an entire rat kidney cortex. Notable classes: normal tubuli (orange), atrophic/dilated tubuli (green/red), glomeruli (pink), abnormal/sclerotic glomeruli (dark blue), arteries (light blue), interstitium (not colored).
Sources
- Kalantar-Zadeh, K., Jafar, T. H., Nitsch, D., Neuen, B. L., & Perkovic, V. (2021). Chronic kidney disease. The Lancet, 398(10302), 786–802. https://doi.org/10.1016/s0140-6736(21)00519-5
- Zhao, D., Wang, W., Tang, T., Zhang, Y.-Y., & Yu, C. (2023). Current Progress in Artificial Intelligence-assisted Medical Image Analysis for Chronic Kidney Disease: A Literature Review. Comput Struct Biotechno. https://doi.org/10.1016/j.csbj.2023.05.029
- Wu, C.-C., Md. Mohaimenul Islam, Tahmina Nasrin Poly, & Weng, Y.-C. (2024). Artificial Intelligence in Kidney Disease: A Comprehensive Study and Directions for Future Research. Diagnostics, 14(4), 397–397. https://doi.org/10.3390/diagnostics14040397
- Wagner, L.-A., Tata, A. L., & Fink, J. C. (2015). Patient Safety Issues in CKD: Core Curriculum 2015. American Journal of Kidney Diseases, 66(1), 159–169. https://doi.org/10.1053/j.ajkd.2015.02.343
- Wei Keat Tey, Ye Chow Kuang, Melanie Po-Leen Ooi, & Joon Joon Khoo. (2018). Automated quantification of renal interstitial fibrosis for computer-aided diagnosis: A comprehensive tissue structure segmentation method. Computer Methods and Programs in Biomedicine, 155, 109–120. https://doi.org/10.1016/j.cmpb.2017.12.004
- National Kidney Foundation. (2002). Clinical practice guidelines for chronic kidney disease : evaluation, classification, and stratification. Saunders.
- Hillaert, A., Stock, E., Favril, S., Duchateau, L., Saunders, J. H., & Katrien Vanderperren. (2022). Intra- and Inter-Observer Variability of Quantitative Parameters Used in Contrast-Enhanced Ultrasound of Kidneys of Healthy Cats. Animals, 12(24), 3557–3557. https://doi.org/10.3390/ani12243557
- Ahmad, Z., Rahim, S., Zubair, M., & Abdul-Ghafar, J. (2021). Artificial intelligence (AI) in medicine, current applications and future role with special emphasis on its potential and promise in pathology: Present and future impact, obstacles including costs and acceptance among pathologists, practical and philosophical considerations. A comprehensive review. Diagnostic Pathology, 16(24). https://doi.org/10.1186/s13000-021-01085-4
- Parajuli, S., Clark, D. F., & Djamali, A. (2016). Is Kidney Transplantation a Better State of CKD? Impact on Diagnosis and Management. Advances in Chronic Kidney Disease, 23(5), 287–294. https://doi.org/10.1053/j.ackd.2016.09.006
- Hermsen, M., Bel, T. de, Boer, M. den, Steenbergen, E. J., Kers, J., Florquin, S., Roelofs, J. J. T. H., Stegall, M. D., Alexander, M. P., Smith, B. H., Smeets, B., Hilbrands, L. B., & Laak, J. A. W. M. van der. (2019). Deep Learning–Based Histopathologic Assessment of Kidney Tissue. Journal of the American Society of Nephrology, 30(10), 1968–1979. https://doi.org/10.1681/ASN.2019020144
- Hermsen, M., Ciompi, F., Adefidipe, A., Denic, A., Dendooven, A., Smith, B. H., van Midden, D., Bräsen, J. H., Kers, J., Stegall, M. D., Bándi, P., Nguyen, T., Swiderska-Chadaj, Z., Smeets, B., Hilbrands, L. B., & van der Laak, J. A. W. M. (2022). Convolutional Neural Networks for the Evaluation of Chronic and Inflammatory Lesions in Kidney Transplant Biopsies. The American Journal of Pathology, 192(10), 1418–1432. https://doi.org/10.1016/j.ajpath.2022.06.009
- Nezami, B.G., Farris, A.B. (2016). Assessing Fibrosis in Kidney Biopsies. In: Patel, V., Preedy, V. (eds) Biomarkers in Kidney Disease. Biomarkers in Disease: Methods, Discoveries and Applications. Springer, Dordrecht. https://doi.org/10.1007/978-94-007-7699-9_51
- Roufosse, C., Simmonds, N., Clahsen-van Groningen, M., Haas, M., Henriksen, K. J., Horsfield, C., Loupy, A., Mengel, M., Perkowska-Ptasińska, A., Rabant, M., Racusen, L. C., Solez, K., & Becker, J. U. (2018). A 2018 Reference Guide to the Banff Classification of Renal Allograft Pathology. Transplantation, 102(11), 1795–1814. https://doi.org/10.1097/tp.0000000000002366
More Blogs
-
Navigating the IVDR: How Aiosyn prioritized compliance from day one
26 May, 2025 • By Diana Rosentul
Read more -
Case Study: Automating Radboudumc’s digital slide Quality Control with AiosynQC
19 December, 2024 • By Anna Correas Grifoll
Read more -
Case Study: Leveraging AI for quantitative assessments of kidney health in diabetic animal models
12 December, 2024 • By Anna Correas Grifoll
Read more -
AI Act and Digital Pathology: Navigating New Regulations
25 September, 2024 • By Anna Correas Grifoll
Read more -
Behind the scenes of pathology AI innovation – Blazej Dolicki’s insights into training, validation, deployment, and beyond
18 June, 2024 • By Victoria Grosu
Read more -
A short overview of the history of pathology: origins, early days, and the transition to novel technologies
16 May, 2024 • By Anna Correas Grifoll
Read more -
The benefits of AI implementation in mitotic figure counting
11 April, 2024 • By Victoria Grosu
Read more -
Diana Rosentul explains the complexities of regulatory compliance for integrating AI-based solutions in the healthcare industry
25 January, 2024 • By Diana Rosentul
Read more